It is the very error of the moon; She comes more nearer earth than she was wont, And makes men mad.
William Shakespeare – Othello

This post is about the recent book Artemis (2017) by Andy Weir which is a novel set on the first permanent Moon station. called “Artemis”. I was looking forward to reading Artemis because I had enjoyed reading his book The Martian. I loved his unorthodox writing style in that book, which fitted in so well with the unfolding of the timeline of the story.
I loved the movie The Martian (2015), directed by Ridley Scott, made from that book. Matt Damon was brilliantly cast as the interplanetary castaway Mark Watney. I loved that it was sci-fi for the near-future. It was tantalising because, with the book and movie, I could easily visualise the colonisation of Mars unfolding along similar lines. It was just so scientifically grounded and believable.
When I started reading Artemis the thought that it was an inspired idea that an African country such as Kenya could take advantage of the technology of moon colonisation to boost their economy to the first-world standard. It was such an optimistic and hopeful view of a world that might be. I loved the idea that the protagonist was an anti-heroine, Jasmine “Jazz” Bashara from Saudi Arabia. That also seemed, at least at first, to be inspired. But later, when she transforms from a small-time smuggler to a big-time crime-boss, still with her smart, way-too-cool sassiness, the characterisation of Jazz losses its credibility and her role just becomes plain silly. Indeed, the whole story, after being so promising, losses its way and degenerates to silliness as I’ll explain.
The colonisation of the Moon
Warning spoilers from this point. Because a large part of the cost of items on Artemis is transportation from earth, the unofficial currency is the SLG for a “soft landed gram” called a slug, denoted as ğ. One slug is one gram of cargo transported by the Kenyan Space Corporation (KSC). Who knew that, owing to the high costs of transportation, that there would be a market in recycled condoms on a moon city?
Weir has a lot of fun with how the laws of physics work on the moon. Turns out there’s a good reason for all those recycled condoms because all the new possibilities for sex in low-gravity is a major tourist attraction for Artemis. Perhaps, even rivalling the attraction of the Apollo 11 visitor centre where Armstrong took his first steps on the lunar surface.
Jazz lives in the poorest parts of Artemis, she’s the estranged daughter of her father, who is a welder, and is herself a porter for delivery of goods. This earns her enough slugs live in a “slab” which is a space only big enough to sleep in and she shares a toilet and shower, down the hallway with others in the same meagre accommodations. She often eats “gunk” which is food source produced in algal vats. Because of her circumstances, she earns extra slugs from smuggling items such as cigars to wealthy businessmen. This way she hopes to save her slugs and provide herself with a better future.
Cigars are strictly forbidden on the moon colony because Artemis has followed the life-support practices of the old Apollo missions by having an atmosphere of pure oxygen at ⅓ rd earth atmosphere (sea level). This avoids the need to transport nitrogen gas cylinders from earth. The pure oxygen atmosphere means that naked flames of any kind are strictly forbidden because any kind of fire would quickly spread throughout the whole city complex.
For this reason, most lunar accommodations are devoid of kitchen facilities. Cooking is performed in special facilities with their own life-support systems that are isolated from the rest of Artemis. Some rather seedy, but wealthy businessmen, can afford to have accommodation with separate life support systems – so that they can smoke cigars whenever the need arises. Her smuggling activities bring Jazz into contact with such people.
A Promising Premise
When Artemis was first built by the KSC there was the need for aluminium for construction purposes and to provide a combustant for solid-fuel rocket engines. So the KSC signed a perpetual agreement with the Sanchez smelter corporation for aluminium production using the mineral anorthite which is plentiful on the lunar surface. In this agreement, Artemis would provide Sanchez with electricity at no cost from their two nuclear power plants which produce excess electricity to the needs of Artemis itself.
In return, this agreement stated that Sanchez would provide Artemis with oxygen at no cost in exchange for the electricity. The oxygen was a by-product aluminium smelting and was piped directly from the Sanchez facility to Artemis. Another by-product of smelting anorthite is silica, which was used by Queensland Glass Manufacturing company to produce the glass required for the construction of Artemis.
These arrangements were essential in the early days of building Artemis but as Artemis has grown they’re out-lived their economic necessity. This means that Artemis desperately needs a new export-based income stream on which to base its economy. It’s at this point the cigar-loving business associate of Jazz, with plans for the mysterious substance known as “ZAFO” [1], comes into the picture.
But there’s a really big problem to overcome first. The uneconomic Sanchez operation has become a front for the South American mafia through which to launder large amounts of ill-gotten funds, free from the attention of authorities on earth. This is where small-time smuggler Jazz Bashara finds herself right in the middle of everything; hunted by both the Artemisian security, as well as a hitman, working for the South American mobsters.
Failing Flat (on the Moon)
It’s at this point, in order to stop the flow of oxygen and invalidate the contract between Artemis and Sanchez, that the all-so-promising premise of the Artemis novel comes crashing down in a heap. You see, Jazz, and her co-conspirators, plot to cause an explosion in the Sanchez plant, In order to get the employees to clear out first, she releases chlorine gas from a cylinder that she has brought with her.
The chlorine sets off the alarms, that warn the employees to clear out, as she expects (all excepting the owner, Loretta Sanchez). But she hadn’t accounted for a cylinder of methane that happens to be standing idly by, and in the explosion that follows, they accidentally produce chloroform which, as everyone knows is a knock-out gas, anestheses the entire population of the moon colony into unconsciousness.
Everyone excepting for Jazz, Loretta and her other associates, who are wearing breathing tanks. Jazz takes this opportunity to escape and switch the supply of oxygen for Artemis to oxygen cylinders that have been stockpiled by her former cigar-loving business associate, now dead by mobster hitman.
It’s a race against time for Jazz and her friends to restore the oxygen supply to revive the anesthetized population before their chloroform exposure becomes lethal.
Oh dear! Oh dear! Andy, why didn’t you call me?
I would have been able to set you straight. Would that it was still possible to run about and grab copies, of the printed and e-book Artemis, from Amazon and the other bookstores, take them to the dark side of the moon and hide them all in a deep deep crater.
There are at least 2 very good scientific reasons why none of what’s been proposed above in the Artemis novel could ever work:
- from medical and toxicological perspectives, the effects of chloroform are not what is described.
- from a chemical perspective, the required amount of chloroform could never be produced as it is described;
These problems are serious for an author that has based his reputation as a sci-fi writer on his ability to portray detailed and accurate science.
Medical and Toxicological Perspectives
Definitive lethality levels of chloroform are difficult to evaluate reliably. Although it is highly likely that exceedingly high levels of chloroform will result in narcosis and subsequent death, the threshold AEGL-3 levels [2] for humans are not available or are too unreliable to be useful. Published AEGL-3 levels for chloroform are based on studies with mice [3]. The published AEGL-3 level for chloroform is 16,000 mg/m3 [3] (which is essentially the same as the 15,000 mg/m3 mentioned in the Artemis book for both the concentration necessary for inducing spontaneous anesthesia and for lethality, after 1 hr duration).
The reason why AEGL-3 data for chloroform is unreliable is the variable response that humans demonstrate when exposed. Humans are able to withstand very high levels of chloroform for short-times for the purpose of medical anesthesia without significant ill-effect. Although chloroform has been replaced with safer inhalation anesthetics, it is still used in special circumstances.
In surgical anesthesia, levels of chloroform up to 25,000 mg/m3 are used for 2-3 minutes and then carefully lowered to 10,000 mg/m3, or less, for the remainder of the surgery. There are good reasons why inhalation anesthetics need to be administered by highly trained medical anesthesiologists.
For the circumstances described in the novel, mass anesthesia by chloroform is likely to see people inadvertently fall in ways that will block their airways and see them suffocate themselves. Indeed, the primary tasks of an anesthesiologist in surgery is to ensure that the patient is positioned so as to keep their airways clear and to monitor their breathing.
There is an article from the annotations section of the medical journal The Lancet of 1865 [4] that is highly appropriate at this point. It seems that with Artemis, Andy Weir has reintroduced a genre of writing that should have been left back in 1865, or back in TV programs of the 60s and 70s. The Lancet article is titled “Chloroform Among Thieves” proceeds with acerbic satirical wit, as highlighted in the quotation below:
The thieves have, it seems, “interrogated nature” with somewhat greater success than has attended the efforts of our best chemists … The common highwayman is an object of our scientific envy … that a highwayman can, by shaking a handkerchief impregnated with chloroform under the nose of his victim, produce instantaneous insensibility.
It seems that even after 150 years, writers haven’t learnt that anesthesia with chloroform is much more difficult than it first appears; it requires much more chloroform, for longer periods, than they think. Likewise, the difference between anesthesia and death is much less than any untrained person expects — which is why it should only be carried out in carefully controlled medical facilities under expert care.
At this point, it has been assumed that Jazz has been able to accidentally produce sufficient chloroform to actually cause anesthesia (or lethality) but this might be an unjustified assumption, as will be examined in detail in the next section.
Chemical Perspective
It’s true that in the Sanchez plant there is chlorine, methane and heat-energy from Jazz’s tampering with the temperature regulation mechanism. It’s quite likely that some chlorine radicals would be generated (see below) to start the initiation phase for a chain reaction. Only relatively few chlorine radicals are needed.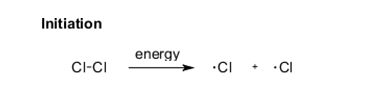
The energy to form chlorine radicals (⋅Cl) is often provided using UV-light, but heating in the range 400-500°C would work just as well.
Highly reactive chlorine radicals, thus generated, start the propagation stage of a chain reaction which is described in detail in a separate support article to this post. Briefly, the gaseous methane and chlorine start to combine in a 4-step chain reaction that proceeds as follows:
- methane –> chloromethane
- chloromethane –> dichloromethane
- dichloromethane –> chloroform
- chloroform –> carbon tetrachloride
So far so good, in the novel Artemis Andy Weir has Loretta Sanchez say essentially the same thing (excepting for omitting the part about the formation of highly toxic and carcinogenic carbon tetrachloride).
The fundamental tenet of industrial chemistry is the law of conservation of matter: we can’t just have matter pop out of nowhere for no reason — work of fiction or not. But this is exactly what happens. It’s one of the main problems with the novel Artemis.
In the novel, with Jazz at the Sanchez smelter, there is a credible mechanism for producing chloroform but we have only one cylinder of chlorine and one cylinder of methane. Furthermore, Jazz has to be able to easily transport that gas cylinder, along with other cylinders of acetylene, oxygen and neon [5] for the welding work that she performs when she sabotages the plant.
The next section can be SKIPPED, it is essentially a tutorial on chemistry and can be skipped on first reading if you prefer.
To make this scenario more realistic, consider a type-Q stainless-steel cylinder of chlorine from Praxair that has a gross mass of 47.7 kg and contains 18.18 kg of compressed liquid chlorine. We’ll also make a reasonable assumption that the methane cylinder, already at the Sanchez plant, is larger than the chlorine cylinder that Jazz brings with her. This way the chlorine is the yielding limiting reagent. We can calculate the amount of chloroform produced as below (using MathCad software from PTC).

There’s 7.7 kg of chloroform that could be maximally produced from a one reasonably sized cylinder of chlorine. Not much when you consider that it has to be spread around the whole city complex.
How much volume does the Artemis complex occupy? The maps shown in the front of the book show 5-domes, consisting of Aldrin, Bean, Collins and Sheppard (all 200 m in diameter), radiating like spokes around the central Armstrong dome (100 m in diameter). The Sanchez plant is connected to the Artemis complex via a 1-km oxygen supply pipeline, it also consists of a dome of 25 m in diameter.

Remembering that the volume of a sphere is and that a dome is half of a sphere, the total volume of the Artemis is around 1.1 million m3. We can now proceed to calculate the airborne concentration:

Skip to here.
The result of the calculation is 7.1 mg/m3, this being the case we would need 2000 cylinders of chlorine to get to a concentration near to the 15,000 mg/m3. In the novel, this is what Loretta Sanchez tells Jazz is the level would be required to induce anesthesia in the good citizens of the moon. From the calculations here, it’s simply impossible to produce enough chloroform to produce the effects described in the book Artemis. with a single cylinder of compressed chlorine — unless you break the law of conservation of matter!
What hasn’t been taken into account in the above equations is that oxygen is also present. In the Artemis novel, Jazz circumvents plant safety measures so that the temperature will reach the melting point of stainless steel at about 1450°C. Thereby destroying the Sanchez plant in an explosion when the stainless steel containment vessel fails.
 In the presence of oxygen, at around 1460°C, the chloroform, and any other alkyl chlorides formed, would simply combust to produce carbon dioxide, water vapour and hydrochloric acid (HCl) vapour. In addition, burning chloroform in oxygen will produce significant traces of phosgene gas (see on RHS).
In the presence of oxygen, at around 1460°C, the chloroform, and any other alkyl chlorides formed, would simply combust to produce carbon dioxide, water vapour and hydrochloric acid (HCl) vapour. In addition, burning chloroform in oxygen will produce significant traces of phosgene gas (see on RHS).
Phosgene is extremely toxic in confined spaces, as little as 3 – 10 mg/m3 would cause death in some of the population if they were exposed for 2 or 3 hours. A point of confusion is that it’s hard to get a mental picture because of the scales involved: chloroform requires macroscopic levels of around 15 g/m3 to cause anesthesia (and possible lethality if this level is sustained for an hour or more). On the other hand, phosgene is far more bioactive (∼1000 times) and its lethal effects can occur at trace levels of 3 – 10 mg/m3 (for exposure of a few hours).
Thus, from the considerations here, it’s more likely that the population would die, or suffer grave injury from phosgene poisoning than undergo mass anestheses.
Final Comments
For a sci-fi writer that has built a reputation on hard and accurate science, Artemis could a backward step for Andy Weir. But life is about learning. He has shown uncommon courage in taking risks to bring hard science to a wider audience. Artemis has a lot going for it and deserves serious attention for the imagination that it shows for what it might be like to live in a city on the moon.
The main problem with the book is in introducing chloroform as a mass knock-out gas for the citizens of the Artemis. This plot device mass anestheses shouldn’t ever be seen again in a sci-fi novel that has any claims to being based in actual science. It’s the kind of impractical science that Jonathon Swift (in Gulliver’s Travels) would have satirically described as “extracting moonbeams from cucumbers.”
Hopefully, this is just an unfortunate aberration from Andy Weir and that with his next book he’ll be back to the same standards he had set with The Martian. Let’s just blame it on the moon this time.
Edit: 19 Jan 2019
Yes, there is an Artemis movie in the works, see at Unbound Worlds.
Edit: 20 Jan 2019
Just got an email from Andy Weir. “Bummer!” he says and adds that the problem is actually worse than I stated because the moon domes are actually full spheres in Artemis (rather than half-spheres as I assumed).
______________
[1] ZAFO stands for “zero attenuation fibre optic” is constructed from a new form of glass core that offers zero transmission loss and can only be constructed economically on the moon. In the novel Artemis, it’s seen as an important export material that will provide future economic stability for the lunar colony.
[2] Acute exposure guideline levels (AEGL) are a way of expressing human health effects from rare exposures to airborne chemicals. AEGL-3 is the airborne concentration (expressed as mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
[3] Committee on Acute Exposure Guideline Levels; Committee on Toxicology; Board on Environmental Studies and Toxicology; Division on Earth and Life Studies; National Research Council, Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 12, Washington (DC): National Academies Press (US). Published: 2012 Apr 27; available online; accessed: 18th November 2018.
[4] Medical Annotations, The Lancet, 86, 2200, p 490-3; 1865.. Available online accessed: 18th November 2018.
[5] Argon is used for welding on earth because it’s plentiful (it can be extracted from liquid air). On the moon, neon is preferable because it weighs about half that of argon for the same amount.


I said the same thing about Harry Potter! In all seriousness great post. Sad, this story sounds like it had great potential. Ill still read it and tbh I probably wouldn’t of questioned the anesthesia part of the story… like chemical magic in the futures w/e. BUT good guys setting off large scale explosions after you have already established small tight living spaces – in high oxygen environment. Questionable even to me.
I was genuinely enjoying the book up until about 2/3rds the way through. Then the chloroform knock-out gas plot device really rattled my cage.
Sounds like a good book, I enjoyed the Movie Martian. Still reading Aliens.